



Founded in 2000, Cyclopharma is specialized in the development and the commercialization of molecular imaging solutions, capable of characterizing diseases and adapted to individual patients.
With more than 100 collaborators and a turnover of 20 million Euros, Cyclopharma is investing significantly in collaborative R&D. Building upon its extensive network in France, Cyclopharma has initiated its international development, with the objective of becoming an European leader in personalized medicine.

As a member of the Pharm’image EIG, Cyclopharma brings its know-how and experience in R&D platform management, to commercialize and promote the expertise, tools, and multi-disciplinary competencies of all the members and partners within the consortium.



Founded in 1995, Oncodesign is the European leader in the preclinical evaluation of anticancer therapies and expert in drug and biomarker discovery. Leveraging its valuable experience and expertise, from working with more than 600 clients and partners including most of the major pharmaceutical companies, along with its comprehensive technological platform combining state-of-the-art medicinal chemistry, advanced cellular pharmacology and animal modelling, identification of therapeutic resistance mechanisms and medical imaging, Oncodesign is able to predict and identify each molecule’s therapeutic usefulness and potential to become an effective drug.
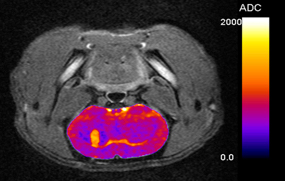
Being at the center of the Pharm’image initiative, as the industry authority in pharmaco-imaging for oncology, Oncodesign leads the IMAkinib consortium, launched in 2009 and financially supported by BPI-France, in collaboration with Cyclopharma laboratories and Ariana, with the goal to identify and develop novel PET radiotracers specifically for targeted therapies.
Oncodesign is an expert in translational research with extensive experience in preclinical experimentation, and the development of innovative imaging biomarkers for new targeted therapies as well as treatment follow-up in the clinical setting.



The Centre Georges-François Leclerc (CGFL) is the comprehensive anti-cancer center of the region of Burgundy. This private non-profit organization is specialized in oncology and has several missions: healthcare, teaching, and research. The CGFL is the only accredited CRC (Clinical Research Center) which is ISO 9001-certified in Eastern France.
Through its integrated technology platforms, CGFL is at the forefront of oncology research, ranging from basic to translational research, encompassing molecular biology, functional, and molecular imaging, and also clinical and epidemiological research.
 The CGFL lends its preclinical and clinical imaging technical expertise and translational research capabilities to the Pharm’image consortium.
The CGFL lends its preclinical and clinical imaging technical expertise and translational research capabilities to the Pharm’image consortium.
A complete set of preclinical imaging modalities are available within the CGFL preclinical imaging platform, which belongs to the nuclear medicine department and enables comprehensive translational expertise from preclinical research to clinical development.
To allow its patients access to innovative therapeutics, the CGFL places emphasizes on clinical research, where close to 20% of patients are enrolled in clinical trials. The clinical investigation center has an early phase unit which conducts phases 0/1 trials, while the clinical research department runs targeted and large-scale phases 2/3 trials.



CheMatech, founded in 2005 based on the know-how from the Institute of Molecular Chemistry at the University of Burgundy, is the European leader in the design and synthesis of chelating agents for nuclear medicine and molecular imaging.

As a co-founder of Pharm’image, Chematech contributes its expertise in radiometal chelation. CheMatech provides the most appropriate chelating agent and tailored solution for a given radioisotope.



NVH Medicinal is a biotech company based in Dijon dedicated to the development of engineered protein-based products for the diagnosis and treatment of collagen related-diseases. NVH develops recombinant collagen-like proteins belonging to a new class of engineered protein, referred to as “recombinamers”, with potent applications in hemostasis and regenerative medicine. Moreover, leveraging its expertise in protein engineering, NVH Medicinal is also a customized services provider in the field of biologics.
 Located in Dijon, Engicare® is a new and integrated platform created through the collaboration between NVH Medicinal® and the preclinical imaging platform of the CGFL at Dijon in Burgundy. Its strong experience allows Pharm’image to benefit from advanced customized research services, from protein conception to biological validation. For example, the development of antibodies and derived recombinant therapeutic to imaging application like tracer. http://www.engicare.fr/fr/
Located in Dijon, Engicare® is a new and integrated platform created through the collaboration between NVH Medicinal® and the preclinical imaging platform of the CGFL at Dijon in Burgundy. Its strong experience allows Pharm’image to benefit from advanced customized research services, from protein conception to biological validation. For example, the development of antibodies and derived recombinant therapeutic to imaging application like tracer. http://www.engicare.fr/fr/



As the premier hospital in the Burgundy region, the CHU is also ranked in the top 20 of best hospitals in France. The patient is at the heart of the organization.
The CHU Dijon Bourgogne as well as all of its employees work every day to deliver treatments of quality to all of its patients.
Devotion and excellence for everybody are our core values.
Innovation and research
Developed as an university-hospital network, the CHU Dijon Bourgogne adopts a constant innovation approach. Its research activities allows clinicians to better understand, prevent, diagnose, and treat diseases. As part of its educational responsibility, the facility also manages several institutes and schools that are in charge of training paramedics and midwife professionals. Moreover, it works closely with the University of Burgundy for the training of medical students.
Our assets
The CHU Dijon Bourgogne has one of the most efficient and up-to-date medico-technical infrastructure to offer the best conditions for quality care:



The General Hospital of Nevers is organized with six medical and medico-technical activity centers. More than 1,800 employees use their expertise and skills in order to offer high-quality patient care. Jean-Michel SCHERRER is the head of the General Hospital of Nevers which can offer more than 900 beds.
The nuclear medicine department
The CHAN has two SPECT/CT cameras and one TEP/CT camera within its nuclear medicine department. The CHAN develops its collaboration thanks to partnership agreements with the CGFL. The aim is to improve the quality of service and help the collaboration between both nuclear medicine departments.
The nuclear department has also developed a radiotherapy partnership with a VPN connection as well as a partnership with the physical department of Blaise Pascal University in Clermont Ferrand.